 « Click here to return to The MTM Review. PRAXBIND is a humanized monoclonal antibody fragment (Fab) indicated in patients treated with Pradaxa when reversal of the anticoagulant effects of dabigatran is needed: For emergency. Jennings added that, since about 1% to 4% of users are likely to experience major bleeding episodes and another 1% of the group is likely to undergo emergency surgery, as many as 500,000 patients will require, within 5 years, drugs that can rapidly reverse anticoagulation. Jennings estimated that 23 million to 36 million patients in the developed world could be using the new anticoagulants by 2020. health regulators approved Praxbind, a reversal agent made by Germany Private held Boehringer Ingelheim, for use in emergency situations by patients.  
The sponsor’s proposed indication is as follows. The special presentation, which occurred before the FDA approval of Praxbind was announced, was led by Lisa Jennings, PhD, professor and director of the Vascular Biology Center at the University of Tennessee Health Science Center in Memphis. BLA 761025 supporting document 2 letter date Februfor Praxbind® (idarucizumab, BI 655075) should be granted accelerated approval from a clinical perspective. A presentation recently at the annual Transcatheter Cardiovascular Therapeutics meeting suggested that the approval of reversal agents could increase use of the new anticoagulants for stroke prevention in patients with atrial fibrillation.  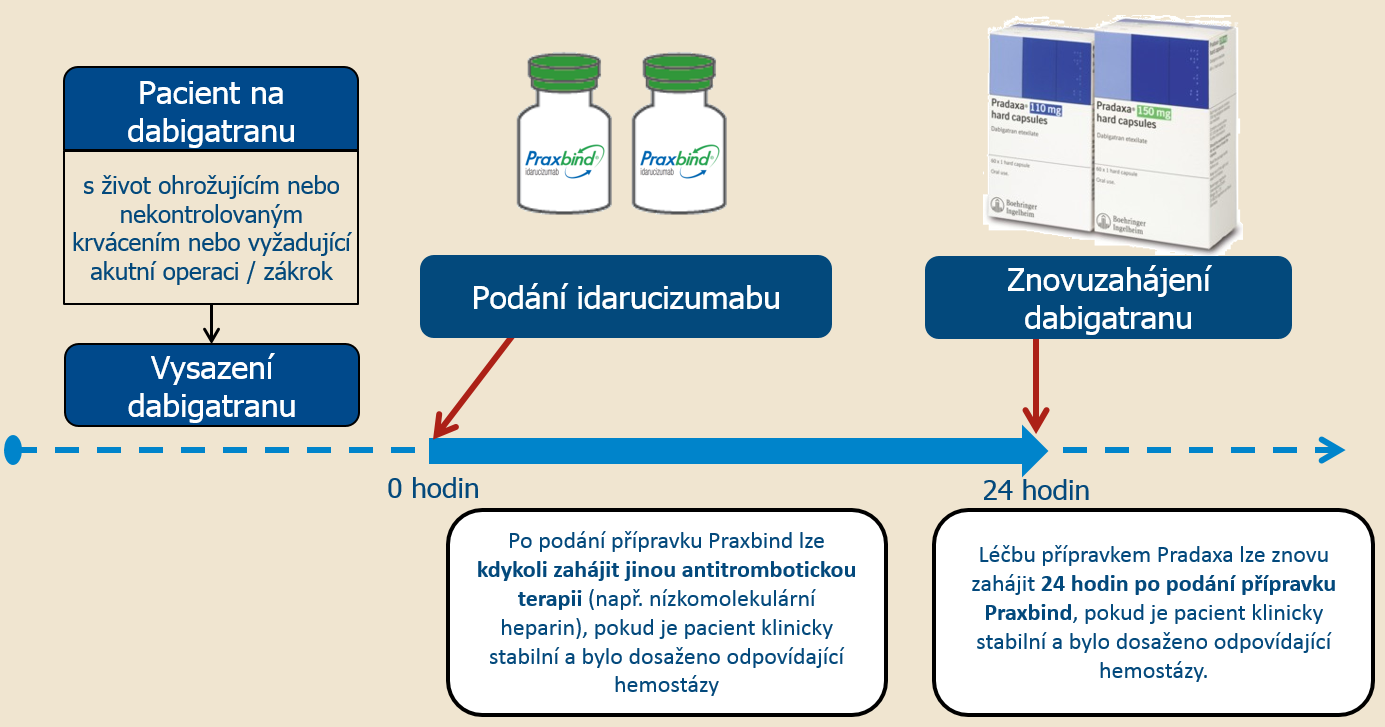
Washington, DC-The FDA recently accelerated approval of an agent, Praxbind (idarucizumab), to reverse Pradaxa’s blood-thinning effects in an emergency situation. The FDA granted accelerated approval to Praxbind ®, a reversal agent (antidote) to Pradaxa ®. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
